-
Project IDG2015-210
-
RFP Year2015
-
Awarded Amount$964,500DiseaseMalariaInterventionDiagnosticDevelopment StageProduct ValidationCollaboration PartnersJuntendo University , Kenya Medical Research Institute (KEMRI-CGHR) , Malaria No More Japan (MNMJ) , National Institute of Advanced Industrial Science and Technology , Institute of Tropical Medicine (NEKKEN) Nagasaki University , Panasonic CorporationPublication
Introduction and Background of the Project
Introduction
Light microscopy is widely used for malaria diagnosis. Stained parasites are directly observed and counted under a microscope; however, the method requires considerable preparation time and skills. Nowadays, the number of skilled technicians is not enough, and remote health facilities seldom have them. The method also requires a stable light source. Electric light is an ideal source for the method, but stable electricity is often unavailable in malaria endemic areas. On the other hand, rapid diagnostic test (RDT) kits are increasing popular in these days because of their simplicity and cost performance. The disadvantages of RDT are a qualitative result and frequent false positives. Therefore, the currently available RDTs should not be used for monitoring patients treated with drugs. Moreover, asymptomatic malaria infection is an obstacle to controlling and eliminating malaria, because the majority of asymptomatic cases are under the detection limits by microscopy and RDT. Molecular methods such as PCR (polymerase chain reaction) are sensitive enough to detect most asymptomatic cases, but the procedure is time-consuming and not cost effective. Currently we are developing a new diagnostic system to overcome these problems. The prototype of this system has already showed greater performance compared to the conventional methods. The system is ready for further improvement through laboratory and field tests in malaria endemic areas.
Project objective
A majority of asymptomatic malaria infections is under the detection limit by microscopy and/or RDT (Fig. 1). Although RDTs are useful for first line screening to support treatment, they should not be used for monitoring the effectiveness of treatment and an elimination program because of the low sensitivity and specificity. The main project objective is to develop a simple and fast malaria diagnostic system with high performance. Health workers should be able to use this system without considerable training. This system should be cost effective and operational in remote health facilities. The system can be used not only for diagnosis but also for monitoring patients and eliminating malaria. The specific objectives that we are aiming during this funded period are 1) to integrate multiple functions or components to one system, 2) to increase sensitivity for detecting at least 0.0005% parasitemia (the system will be able to detect infection even though only 0.0005% of red blood cells are infected. We will try to improve it to 0.00005% after this project, which is 100 times greater than RDT. ), 3) to reduce false positives nearly zero by enhancing the quantification technology, 4) to improve the system to handle at least 10 blood samples simultaneously, 5) to make all procedure automated (it should require only a single keyboard or mouse action after applying blood samples), 6) to display summary test results on a monitor within 10 minutes, and 7) to make the system portable and battery operated. .
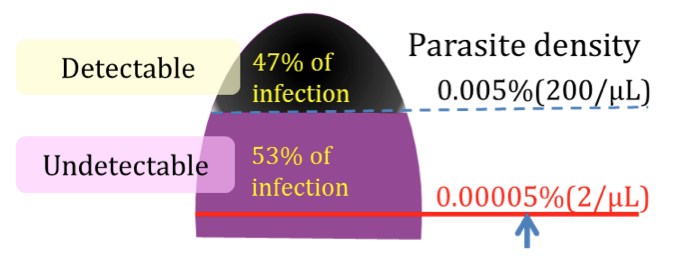
Fig. 1. This new malaria diagnostic system will have high sensitivity.
Project design
This malaria diagnosis system has three main functions or components, filtering red blood cells, spreading red blood cells to a single layer for accurate detection of Plasmodium parasites, and quantifying them in the blood cells. We will integrate these functions to one system. For filtering function, we will design a disposal compact polystyrene container (cell microarray chip) and a compact disc that can host multiple cell chips. As the disc will rotate in a device resembled to a DVD driver, blood will migrate toward a small opening of the cell chip. A filter at the opening will separate only red blood cells from the other blood components, and red blood cells will enter inside the chip. Then, red blood cells will form a single layer and will be stained with fluorescent dyes in the chip. We will also add functions of detecting and quantifying parasites in the blood cells applying the modern digital imaging technology. After integrating these functions mechanically, we will write a software program to automate the procedure. We will also design a function for displaying test results on a monitor. In the lab, we will evaluate this integrated system using infected red blood cells with cultured parasites. Once confirmed that this system works properly and satisfies our numerical targets, we will evaluate this system with blood samples from residents in malaria endemic areas in Africa. Then, we will make the system operational in local health facilities.
How can your partnership (project) address global health challenges?
This system can dramatically improve the current situation of malaria diagnosis and treatment in endemic areas. Microscopy and RDT for routine diagnosis may be replaced with this system. In particular, this will make a great contribution in rural health facilities with an insufficient number of health workers and little equipment. When a health facility receives a large number of patients during a malaria season, the system will help both health workers and patients by handling multiple samples. The superior ability to quantify parasites will lead to accurate diagnoses and proper treatments, which may eventually reduce medical costs. Moreover, this system will be operational with solar energy in an area without electricity. With its portability this system will be also useful for mobile clinics. As this system requires little training, it will become handy for health facilities without skilled microscopists and training budget. Global efforts to combat malaria in the past decade achieved substantial reduction of deaths. Now several endemic countries are targeting malaria elimination. In the era of elimination, malaria prevalence will become extremely low; the number of malaria parasites in a person may become lower than the detection limit of microscopy. On the other hand, individuals with low parasitemia still become a source of parasites for others, and identifying infected individuals is indispensable to achieve malaria elimination. In this regard, this diagnostic system can be best fit for such situation.
What sort of innovation are you bringing in your project?
This diagnostic system is built with a totally different concept from the currently available tools. The system is built with some highly innovative technologies. To improve sensitivity and specificity, this system needs to separate red blood cells from the other blood components. Without the other blood components (or noise), it is able to detect and quantify parasites in the blood cells, which reduces errors such as false positive. The filter invented with SiO2 nano-fibers made this possible. Another innovative future is forming mono-layer of red blood cells. As red blood cells are arranged in a single layer, parasites in the cells will be detected correctly. This requires only 2 μl of blood. For detecting and quantifying parasites in red blood cells, this diagnostic system uses the latest digital image reading technology with a DVD type scan disc. Then, through a computer monitor, health workers actually are able to see malaria parasites in red blood cells. This process will be automated and require less than 10 minutes.

Fig 2. Proposed malaria diagnosis system with a CD type scan disc and an image reader.
Role and Responsibility of Each Partner
Panasonic will be the overall project leader and principle investigator overseeing the progress of the project. Panasonic will also lead the development of blood cells separation device, the cell chip for plasmodia detection, and fluorescent detector. Juntendo University will work with AIST on the development of principle for high sensitive technology and field testing. Juntendo University will also be responsible for discriminate malaria specifics, extract issues for diagnosis, and perform statistical evaluation of field data. National Institute of Advanced Industrial Science and Technology (AIST) will work with Juntendo University on the development of principle for high sensitive technology. AIST will also be responsible for discriminating species of plasmodia and will work on chip PCR technology. Institute of Tropical Medicine at Nagasaki University will work with partners on the evaluation of the technology in the field trials. The Institute of Tropical Medicine will work with partners to manage field trials in Kenya, conduct field test of the diagnostic system and detecting problems and control ethics on site, and conduct preclinical testing. Malaria No More Japan (MNMJ) will provide partners with consultation for field testing and conduct marketing effort in Africa. Kenya Medical Research Institute (KEMRI-CGHR) will work with partners on conducting the field trails and preclinical trials. Kenya Medical Research Institute (KEMRI-CGHR) will also work with partners on detecting problems and control ethics on site.
Final Report
1. Project objective
We develop the malaria diagnosis technology which is comprised of three element technologies: a one-step filter device, a cell chip that realizes quantitative measurement of Plasmodium parasites and a battery operated fluorescent detection device. This diagnosis system is developed integrating three functions, fluorescent measurement, signal processing and quantitative measurement. The integration was realized using a turnkey system; the detection device in the form of optical disc drive was operated with a laptop PC. This system will be validated using, actual blood samples from the field.
2. Project design
Panasonic was a principle investigator. The role of Panasonic was the management of this project. Malaria No More Japan (MNMJ) had a function of consulting for Panasonic, National Institute of Advanced Industrial Science and Technology (AIST), Juntendo University, Nagasaki University, and Kenya Medical Research Institute (KEMRI). Panasonic had a responsibility regarding the development of blood cells separation device, cell chip, and fluorescent detector. The role of AIST was the development of basic technology for high sensitive Plasmodia detection. The role of Juntendo University, Nagasaki University and KEMRI conducted the field evaluation. MNMJ had a marketing for achieving success this project.
3. Results, lessons learned
Integrated the cell microarray chip into the optical disc equipped with filter device
We developed one-step erythrocyte filter device. This device functions with cytapheresis that is based on the size and absorptive effects of leukocytes. We realized removal rate of over 99.9% leukocytes and recover rate of more than 40% of erythrocytes in the blood.
The optical disc that hosts the filter devices also contain multiple units for spreading erythrocytes to monolayer and staining them. Each unit is connected to a filter device with a channel. As the disc spins, the filter device recover erythrocytes, and they migrate toward the unit through the channel with centrifugal force. The monolayer of erythrocytes allow the optical pick up device to detect and quantify parasites in erythrocytes.
We developed a diagnostic device optical disc so that simultaneous multi-specimen diagnosis can be conducted of large numbers of malaria patients.
Development of measurement system
We developed a measurement device operated from a PC. The prototype estimate the number of infected erythrocytes. This prototype connected with a PC that can be operated by batteries at remote health facilities. Our system is constructed basically with the malaria parasite detector, the sensor disc and a PC which was installed with exclusive software for controlling the detector.
A summarized measurement procedure is as follows. (1) A blood sample is collected with a capillary tube, and the blood is diluted with buffer solution. (2) The diluted blood sample is put into the scan disc. (3) A user inserts the scan disc into the detector. (4) The detector captures the fluorescent signal of parasites. (5) The computer calculates malaria infection rate and displays an image of parasites in erythrocytes.
Evaluation of the diagnostic system shall be conducted using blood from malaria patients in regions in Africa and Melanesia that are malaria endemic.
We had a Pre-marketing Intelligence regarding overview of Malaria Market in East Africa. We cleared the target of our system compared with RDT, microscopy, and PCR. The hypothesis of marketing strategy was also proposed in our survey of marketing.
Investment
Details
Development of a fully automated malaria diagnostic system and field evaluation for practical use




